Quality Assurance and Test Automation for Life Sciences and Digital Health
Accelerating High-Confidence Releases for Healthcare Systems
In regulated health environments, every release carries stakes far beyond customer inconvenience. Defects in patient portals, telehealth apps, clinical trial platforms or research systems can impact patient safety and operational efficiency.
Aspiritech embeds human-led QA and automation expertise into your teams to reduce defects, accelerate release velocity, and maintain compliance, all without increasing headcount.
The Challenge: Scaling QA Across Complex Healthcare Systems
Quality assurance bottlenecks are magnified in health technology.
Manual testing slows critical updates for patient portals and research platforms. Regression cycles grow as platform complexity inevitably increases. Limited automation expertise among in-house teams impedes timely releases. Finding and retaining reliable QA professionals becomes a drain on real progress.
When delays or defects risk regulatory compliance and institutional reputation, QA and automation become mission-critical functions in digital health.
The Cost to Fix Bugs Increases in Later Development Stages
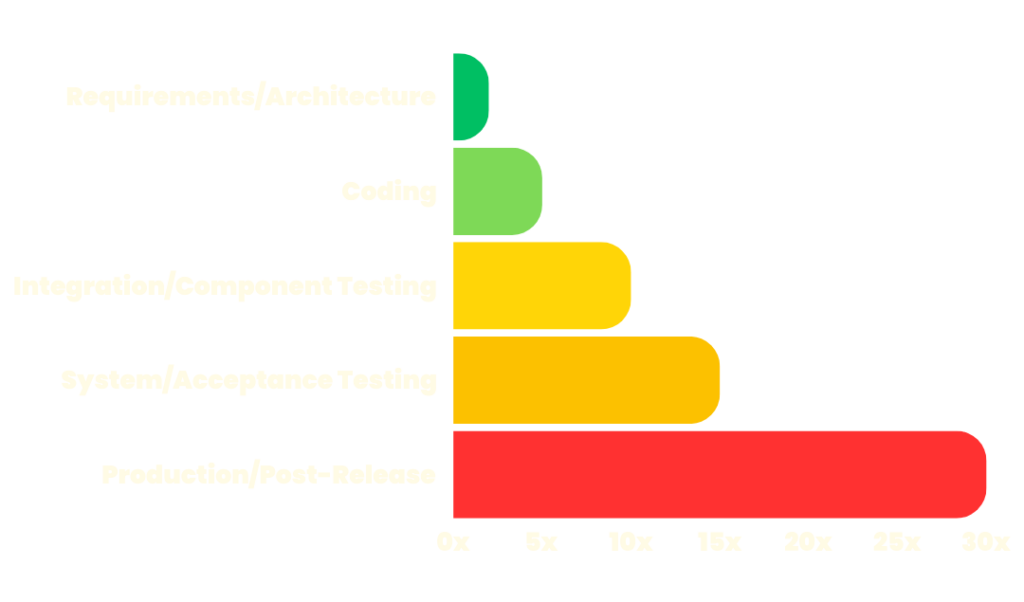
Source: National Institute of Standards & Technology
The effort—and cost—involved in identifying and fixing defects increases exponentially as software moves through the development cycle.
We’ll help your team identify and remediate usability issues in the testing phases, which mitigates costs and prevents major outages and downtime.
The Solution: Human-Led QA That Accelerates Releases
Aspiritech delivers embedded QA and test automation designed for highly regulated digital health environments. Our approach combines:
- Human-led manual and exploratory testing that identifies real-world defects that automated scripts often miss
- Test automation frameworks optimized to reduce backlogs and align with CI/CD pipelines.
- Flexible QA team models to scale with release cycles, whether on-demand, blended, or fully embedded.
By embedding QA directly into workflows, organizations release faster, reduce defects, and maintain regulatory and patient safety standards.
Outcomes That Matter
Life science and digital health teams that work with Aspiritech see measurable impact:
- Accelerated release cycles for patient-facing portals, telehealth app, and research platforms
- Fewer production defects and improved patient and provider experience
- Streamlined regression testing and predictable release schedules
- Optimized QA staffing and reduced reliance on limited internal resources
- Clear ROI from automation investments, freeing teams for higher-value work
With these capabilities, digital health teams gain speed and confidence in every release.

Get Better Quality Assurance Solutions Now
Let Us Know How We Can Help!
We are more than a QA vendor. As a strategic partner for regulated digital health, our human-led approach combines user-centered QA and automation expertise to deliver measurable quality outcomes for patients and institutions.
Send us an email or set up a time to discuss how we can help your team build a platform that exceeds expectations.
Case Study: Ensuring Accuracy and Reliability in Diagnostic Documentation for Abbott
Accuracy and compliance in healthcare documentation require more than careful review. They require structured processes that scale across products and teams.
Abbott Laboratories, a global leader in diagnostics and medical devices, engaged Aspiritech to implement a structured quality assurance approach for Instructions for Use (IFUs) and other critical product documentation.
From Manual Reviews to Scalable QA
While Abbott had skilled teams, manual IFU reviews were time-consuming and prone to inconsistency and delays. Discrepancies could create regulatory risk or compromise clarity for patients and providers. Without a standardized process, review cycles were inefficient and relied heavily on limited internal resources.
Implementing a Structured, Human-Led QA Program
Over the course of the engagement, Aspiritech delivered a centralized, scalable QA process tailored to Abbott’s documentation needs. We executed the following:
-
Conducted detailed side-by-side evaluations of IFUs, checklists and source documents
-
Developed and refined centralized review templates and reporting workflows
-
Verified data accuracy and material consistency across products
-
Embedded flexible, neurodivergent QA teams to scale with project complexity and release cycles
The result was a consistent, audit-ready QA process. Through our accuracy-driven partnership, Abbott now maintains compliant documentation with streamlined workflows and improved efficiency.
Client Testimonials
Abbott
Frequently Asked Questions About Quality Assurance for Pharmaceutical, Biotech and Digital Health Organizations
We work with diagnostic systems, patient portals, telehealth platforms, clinical trial applications and other regulated digital health environments.
Our teams embed directly into your development process, working alongside engineers and product managers to provide manual, exploratory and automated testing without disrupting operations.
Absolutely. Our QA and automation processes are designed for highly regulated environments, ensuring compliance with FDA, HIPAA, and 21 CFR Part 11, as well as other accessibility standards.
Very! We offer on-demand, blended or fully embedded models that scale with your team’s growing needs.
Our neurodivergent professionals excel at precision and innovative creativity. These strengths translate into fewer defects, faster releases and more consistent outcomes for regulated digital health applications.
Depending on project scope, we can deploy dedicated testers within weeks, aligning resources to your timelines while maintaining thorough, high-quality QA practices.
